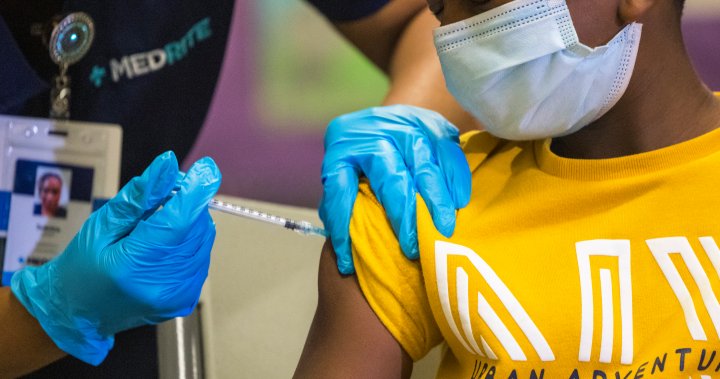
The U.S. Meals and Drug Administration on Monday approved using a 3rd dose of the Pfizer and BioNTech COVID-19 vaccine for kids aged between 12 and 15 years, and narrowed the time for all booster pictures to 5 months from six months after major doses.
The company additionally approved a 3rd shot in youngsters aged 5 by means of 11 years who’re immunocompromised.
The FDA mentioned it reviewed revealed knowledge and actual world proof on the security of booster doses supplied by the Israeli Ministry of Well being together with knowledge from over 6,300 people 12-to-15 years of age who acquired a Pfizer shot.
International COVID-19 circumstances are surging as a result of Omicron variant and well being authorities have warned that its extraordinarily excessive transmissibility might overwhelm many well being methods.
Laboratory checks have proven that two doses of the Pfizer-BioNTech and Moderna vaccines generate low immune responses in opposition to Omicron, whereas boosters seem like protecting in opposition to the highly-mutated variant.
View hyperlink »